Low-Level Laser Therapy for Neuropathy
For people living with burning, tingling, or stabbing nerve pain, noninvasive laser therapy—also called photobiomodulation or “cold laser”—is drawing interest as a drug‑sparing option. It uses low‑energy red and near‑infrared light to influence cellular activity without heating tissue. Here’s how it works, what the evidence shows, and practical tips before you book.
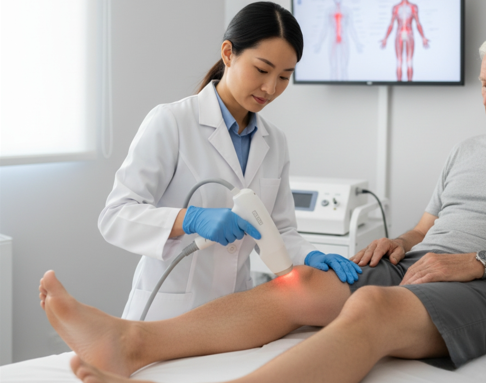
What this therapy is
Low‑level laser therapy (LLLT) for neuropathy delivers red and near‑infrared light to tissues at energies that modulate biology rather than cut or cauterize. The light is absorbed by mitochondria, which can boost ATP production and trigger signaling that may dampen inflammation and oxidative stress [1]. Laboratory and animal studies suggest effects on ion channels, axonal transport, and pathways involved in nerve repair and regeneration [1,2]. In clinics and studies you’ll see terms like photobiomodulation, cold laser therapy, and peripheral neuropathy laser treatment used to describe this approach.
Why it matters now
Peripheral neuropathy is common, particularly with diabetes, chemotherapy, and aging. Painful diabetic polyneuropathy affects a sizable proportion of people with diabetes and often persists despite medication [3,4]. Concerns about long‑term opioid use and variable drug response have prompted interest in FDA‑cleared light devices as noninvasive adjuncts. Early studies report short‑term pain and sensory improvements, but larger, high‑quality trials are still needed to guide routine use [2]. Photobiomodulation is also being explored for chemotherapy‑induced peripheral neuropathy, with promising yet preliminary data [5].
Potential benefits and outcomes
What results can you expect? Outcomes vary by condition, device, and dose. Across small randomized or controlled studies, many patients report meaningful pain reductions—often in the 20–40% range from baseline—and improved vibration or thermal sensation after several weeks versus sham [2,5]. Potential upsides include:
- Possible gains in nerve conduction or small‑fiber function markers
- Minimal downtime and a low reported adverse‑event rate when properly dosed
How does LLLT compare with TENS? TENS can provide short‑term analgesia via electrical stimulation, while photobiomodulation aims to influence cellular processes underlying pain. Evidence for TENS in neuropathic pain is mixed and low‑certainty [10]; photobiomodulation has biologic plausibility and growing, though still limited, clinical support [2,5]. Many clinicians combine them.
How treatment typically works
A visit usually starts with mapping painful or numb areas and reviewing medications and contraindications. The clinician applies light at specified wavelengths—commonly 600–1,000 nm—using energies guided by consensus dosing frameworks such as WALT recommendations [6]. Sessions last 10–30 minutes. Typical protocols schedule 2–3 sessions per week for 4–6 weeks (about 8–15 total), followed by reassessment and possible tapering [6].
Costs vary by market and device. Many clinics charge $50–$150 per session, with package pricing common [8]. Insurance coverage is limited; there is no national Medicare coverage determination, and many insurers still consider LLLT investigational for neuropathic pain, so expect to self‑pay unless your plan specifies coverage [9]. Considering home devices? Prioritize transparent power/irradiance specifications and verify that any “FDA‑cleared” claims match the device’s actual cleared indication; clearances are device‑ and condition‑specific, so review the FDA letter carefully [7].
Risks and common limitations
When provided by trained clinicians, adverse effects are uncommon. Do not look into the beam; eye protection is essential. Avoid treatment over known or suspected malignancy, the uterus during pregnancy, active bleeding, and photosensitive rashes. People on photosensitizing drugs should proceed cautiously. Transient soreness, warmth, or brief symptom flares can occur; tattoos or very dark skin may require lower doses [6]. Importantly, evidence is heterogeneous—devices, wavelengths, and dosimetry vary—making study comparisons challenging [2]. Cold laser therapy is not a cure and should complement, not replace, diabetes management, physical therapy, and guideline‑directed medications for painful diabetic neuropathy [3].
Smart next steps to consider
- Consult a clinician experienced with photobiomodulation for peripheral neuropathy to confirm candidacy and set realistic goals.
- Clarify dose and duration: number of sessions, expected timeline, and when you’ll reassess progress [6].
- Compare options—LLLT, TENS, physical therapy, and topical/oral medications—and consider combining approaches tailored to your symptoms [10].
- Budget ahead. Confirm per‑session costs and any coverage through your insurer or Medicare Advantage plan [8,9]. For home units, favor devices with published data and clear specifications.
- Track outcomes. Use a pain diary and simple sensory checks (e.g., monofilament, vibration) to judge whether treatment is meeting your goals and adjust the plan accordingly